Peripheral T-cell lymphoma (PTCL)| Primary Research (KOL’s Insight) | Competitive Intelligence | Market Analytics & Forecast 2032
Price range: $6,989.00 through $20,967.00
Mellalta’s analysts estimate that the United States market is expected to show rapid growth, mainly attributed to, the launch of Adcetris in the front-line setting and expected approval of the potential therapies in the pipeline during the forecast period (2021-2030). Through this, the market size is set to show an extensive jump and grow at a CAGR of 15.3%
Description
Peripheral T-cell lymphoma (PTCL) remains universally hard to treat in both the frontline and relapsed settings. The International PTCL Project reported 5-year OS for PTCL-NOS, AITL, ALK-negative ALCL, and ALK-positive ALCL to be 32%, 32%, 49%, and 70%, respectively. Clinical studies adding active agents to the CHOP regimen have shown limited efficacy improvements, demonstrating the high unmet need in these patients.
Peripheral T-cell lymphoma (PTCL) is a heterogeneous group of lymphomas representing 5% to 15% of non-Hodgkin’s lymphomas (NHL) in the Western world. It is composed of peripheral (systemic) and cutaneous forms developed from T cells and natural killer (NK) cells. Clinically, these lymphomas are often aggressive. The standard of care for the last 30 years has been cyclophosphamide, doxorubicin, vincristine, and prednisolone (CHOP) with or without additional etoposide (CHOEP). A meta-analysis of the outcome of PTCL treated with anthracycline-based regimens demonstrated a 5-year overall survival of only 36.6%. Attempts have been made to improve on this poor outlook by: (i) adding newer drugs to the CHOP backbone (alemtuzumab, romidepsin, brentuximab vedotin); (ii) intensifying the regimen (DA-EPOCH); (iii) developing alternative regimens (eg, gemcitabine-based combinations).
Peripheral T-cell lymphoma (PTCL) Epidemiology
The most recent WHO 2016 Classification of Tumors of Hematopoietic and Lymphoid Tissues, now recognizes 29 discrete types of PTCL. The most common subtypes account for over one-half of all PTCL cases, including PTCL-not otherwise specified, angioimmunoblastic T-cell lymphoma, and anaplastic lymphoma kinase (ALK)-positive or ALK-negative anaplastic large cell lymphoma (ALCL). PTCL is challenging to diagnose, largely owing to a lack of reliable diagnostic markers and limited clinical experience because of the rarity of the disease, which may lead to patients receiving inadequate or delayed treatment.

Peripheral T-cell lymphoma (PTCL) Market Forecast
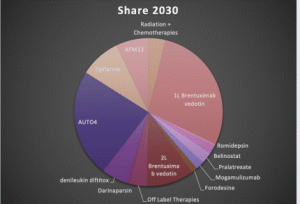
The Peripheral T-cell lymphoma therapy market is expected to experience high growth throughout our study period, estimated to reach $1.9 billion by 2030, representing 15.2 % annual growth. Brentuximab vedotin is already approved for first-line PTCL setting especially in systemic anaplastic large cell lymphoma or other CD30-expressing peripheral T-cell lymphomas (PTCL) [100% CD30 expression rate], including angioimmunoblastic T-cell lymphoma [75% CD30 expression rate] and PTCL not otherwise specified [60% CD30 Expression rate], in combination with cyclophosphamide, doxorubicin, and prednisone (CHP).
KOLs indicated that CD30 expression levels would be the other major factor driving the uptake of Adcetris + CHP. Given the uncommon nature of the disease, research into Cd30 expression hasn’t been extensive—especially as it didn’t impact Chop treatment. Current estimates have placed the level of Cd30 expression somewhere between one-third to ~40% of all PTCL. However, given that the threshold level for Adcetris activity might be very low, with some patients expressing very low levels of CD30, up to 50% of all newly diagnosed individuals could be eligible for treatment.
There is currently no standard therapy for relapsed/refractory PTCL Patients. Despite some PTCL patients having initial responses to chemotherapy, most patients either relapse or fail to achieve remission. A few agents have been approved for relapsed PTCL.
Nevertheless, the response rates for most agents are low and the response durations are not sustained, which leads to eventual relapse. The growth of the ALL market through the forecast period will be driven by emerging therapies Darinaparsin (Solasia), Tipifarnib (Kura Oncology), AFM13 (Affimed), AUTO4 (Autolus), and others. Many new therapies currently in development are expected to be approved during our forecast period of 2022-2032. Emerging therapies are mostly focused on eliminating or reducing unmet needs like the requirements for much more effective treatments for the relapsed/refractory cases of PTCL. Launching these upcoming therapies tends to increase the market size of Peripheral T-Cell Lymphoma (PTCL) in upcoming years.
Report Highlights
- Peripheral T-cell lymphoma (PTCL) Current Market Trends
- Peripheral T-cell lymphoma (PTCL) Current & Forecasted Cases across the G7 Countries
- Peripheral T-cell lymphoma (PTCL) Market Opportunities And Sales Potential for Agents
- Peripheral T-cell lymphoma (PTCL) Patient-based Market Forecast to 2030
- Peripheral T-cell lymphoma (PTCL) Untapped Business Opportunities
- Peripheral T-cell lymphoma (PTCL) Product Positioning Vis-a-vis Competitors’ Products
- Peripheral T-cell lymphoma (PTCL) KOLs Insight
Additional information
| Price | Single User License, 2-3 User License, Site License, Enterprise License |
|---|
Table of Contents
- Peripheral T-cell lymphoma (PTCL) Disease Background
- Peripheral T-cell lymphoma (PTCL) Definition
- Causes & Risk factors
- Symptoms & Staging
- Classification
- Pathophysiology
- Diagnosis
- Conditions Mimicking PTCL
- Peripheral T-cell lymphoma (PTCL) Diagnosis
- Epidemiology Estimated and Forecast to 2032
- Epidemiology Research Method & Data Sources Used
- United States
- Incident Cases of Peripheral T-cell lymphoma (PTCL)
- Relapsed/ refractory Cases of Peripheral T-cell lymphoma (PTCL)
- United Kingdom
- Incident Cases of Peripheral T-cell lymphoma (PTCL)
- Relapsed/ refractory Cases of Peripheral T-cell lymphoma (PTCL)
- Germany
- Incident Cases of Peripheral T-cell lymphoma (PTCL)
- Relapsed/ refractory Cases of Peripheral T-cell lymphoma (PTCL)
-
- Italy
- Incident Cases of Peripheral T-cell lymphoma (PTCL)
- Relapsed/ refractory Cases of Peripheral T-cell lymphoma (PTCL)
- Spain
- Incident Cases of Peripheral T-cell lymphoma (PTCL)
- Relapsed/ refractory Cases of Peripheral T-cell lymphoma (PTCL)
- France
- Incident Cases of Peripheral T-cell lymphoma (PTCL)
- Relapsed/ refractory Cases of Peripheral T-cell lymphoma (PTCL)
- Japan
- Incident Cases of Peripheral T-cell lymphoma (PTCL)
- Relapsed/ refractory Cases of Peripheral T-cell lymphoma (PTCL)
- Current Unmet Needs in Peripheral T-cell lymphoma (PTCL)
- Italy
- Current Treatment Paradigm
- Treatment guidelines for Peripheral T-cell lymphoma (PTCL)
- Regulatory Approvals/Indication and Current Benchmarks
- Current Treatment Chapters
- Belinostat (Onxeo)
- Romidepsin (Bristol Myers Squibb)
- Pralatrexate (Acrotech Biopharma)
- Mogamulizumab (Kyowa Kirin)
- Brentuximab vedotin (Seagen/Takeda)
- Forodesine (Mundipharma International)
- Denileukin diftitox (Eisai)
- Crizotinib (Pfizer)
- Emerging Therapies Chapters
- Darinaparsin (Solasia Pharma)
- Cabiralizumab + Nivolumab (Bristol-Myers Squibb/Five Prime Therapeutics)
- Valemetostat (Daiichi Sankyo)
- AUTO-4 (Autolus)
- Tipifarnib (Kura Oncology)
- Duvelisib (Secura Bio)
- AZD 4205 (Dizal Pharmaceutical)
- Tenalisib (Rhizen Pharmaceuticals)
- Others
- Cost Coverage and Reimbursement plan
- United States Cost Coverage & Reimbursement for PTCL Therapies
- What’s New in 2020
- Future Treatment Paradigm
- Peripheral T-cell lymphoma (PTCL) Competitor Landscape and Approvals Anticipated
- Future Treatment Algorithms and Competitor Positioning
- Key Data Summary for Emerging Treatment
- Total Market Forecast
- Key Summary Findings
- G7 total Market for Peripheral T-cell lymphoma (PTCL) 2022-2032 (USD Million)
- G7 total Market for Peripheral T-cell lymphoma (PTCL) by Therapies 2022-2032 (USD Million)
- Market Forecast by Country
- United States
- United States Market for Peripheral T-cell lymphoma (PTCL) 2022-2032 (USD Million)
- United States Market for Peripheral T-cell lymphoma (PTCL) by Therapies 2022-2032 (USD Million)
- Germany
- Germany Market for Peripheral T-cell lymphoma (PTCL) 2022-2032 (USD Million)
- Germany Market for Peripheral T-cell lymphoma (PTCL) by Therapies 2022-2032 (USD Million)
- France
- France Market for Peripheral T-cell lymphoma (PTCL) 2022-2032 (USD Million)
- France Market for Peripheral T-cell lymphoma (PTCL) by Therapies 2022-2032 (USD Million)
- Italy
- Italy Market for Peripheral T-cell lymphoma (PTCL) 2022-2032 (USD Million)
- Italy Market for Peripheral T-cell lymphoma (PTCL) by Therapies 2022-2032 (USD Million)
- Spain
- Spain Market for Peripheral T-cell lymphoma (PTCL) 2022-2032 (USD Million)
- Spain Market for Peripheral T-cell lymphoma (PTCL) by Therapies 2022-2032 (USD Million)
- United Kingdom
- United Kingdom Market for Peripheral T-cell lymphoma (PTCL) 2022-2032 (USD Million)
- United Kingdom Market for Peripheral T-cell lymphoma (PTCL) by Therapies 2022-2032 (USD Million)
- Japan
- Japan Market for Peripheral T-cell lymphoma (PTCL) 2022-2032 (USD Million)
- Japan Market for Peripheral T-cell lymphoma (PTCL) by Therapies 2022-2032 (USD Million)
- Market Drivers & Barriers
- Appendix
- About Mellalta Meets
- United States
- Key Summary Findings
Sample Enquiry
[wpforms id=”499″]
 English
English 




